Key Structural Differences Between α-Helices and Other Secondary Forms
When I first tried to tell an α-helix apart from other secondary structures, it felt like trying to recognize constellations on a cloudy night—there were shapes and patterns, but no real clarity. The usual textbook definitions about hydrogen bonds and residue counts didn’t quite click. What really tripped me up was not just memorizing facts, but truly understanding why these structures form the way they do—and how that links to what proteins actually do. If you want a deeper dive, check out this comprehensive guide to α-helix structure and function.
Here’s what helped me get past that confusion: secondary structures aren’t just patterns to spot; they’re nature’s clever solutions to physical and chemical challenges. Once you grasp the why behind their shape and bonding, the what and how become much easier to remember and apply.
α-Helices: Nature’s Tight Springs
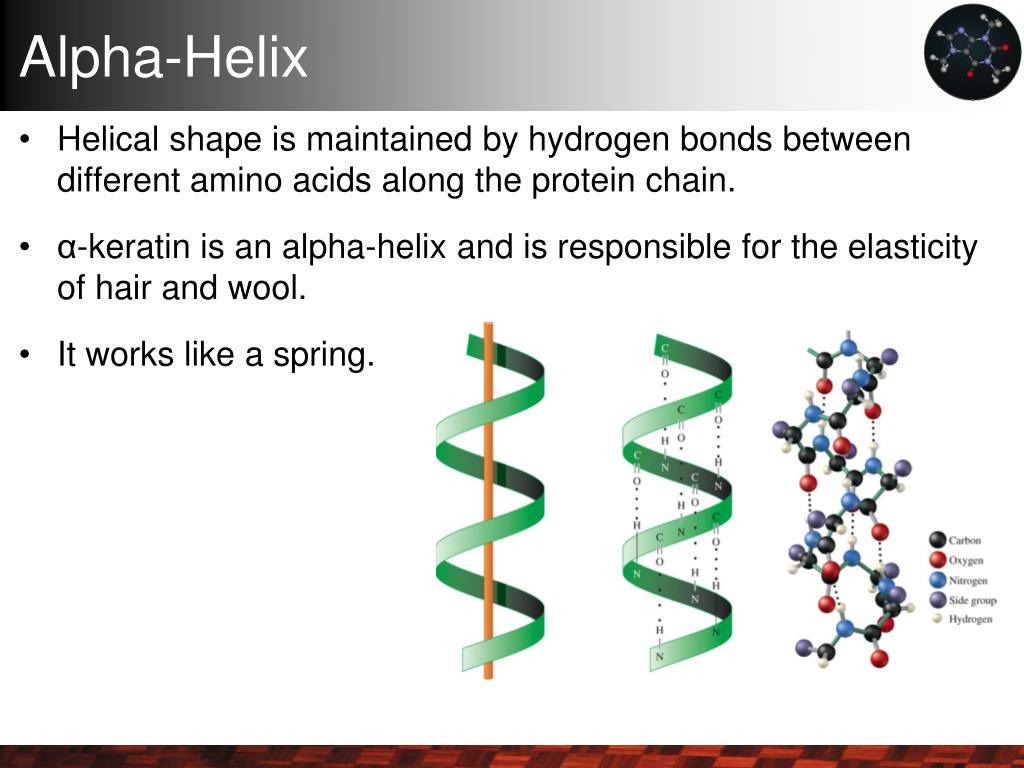
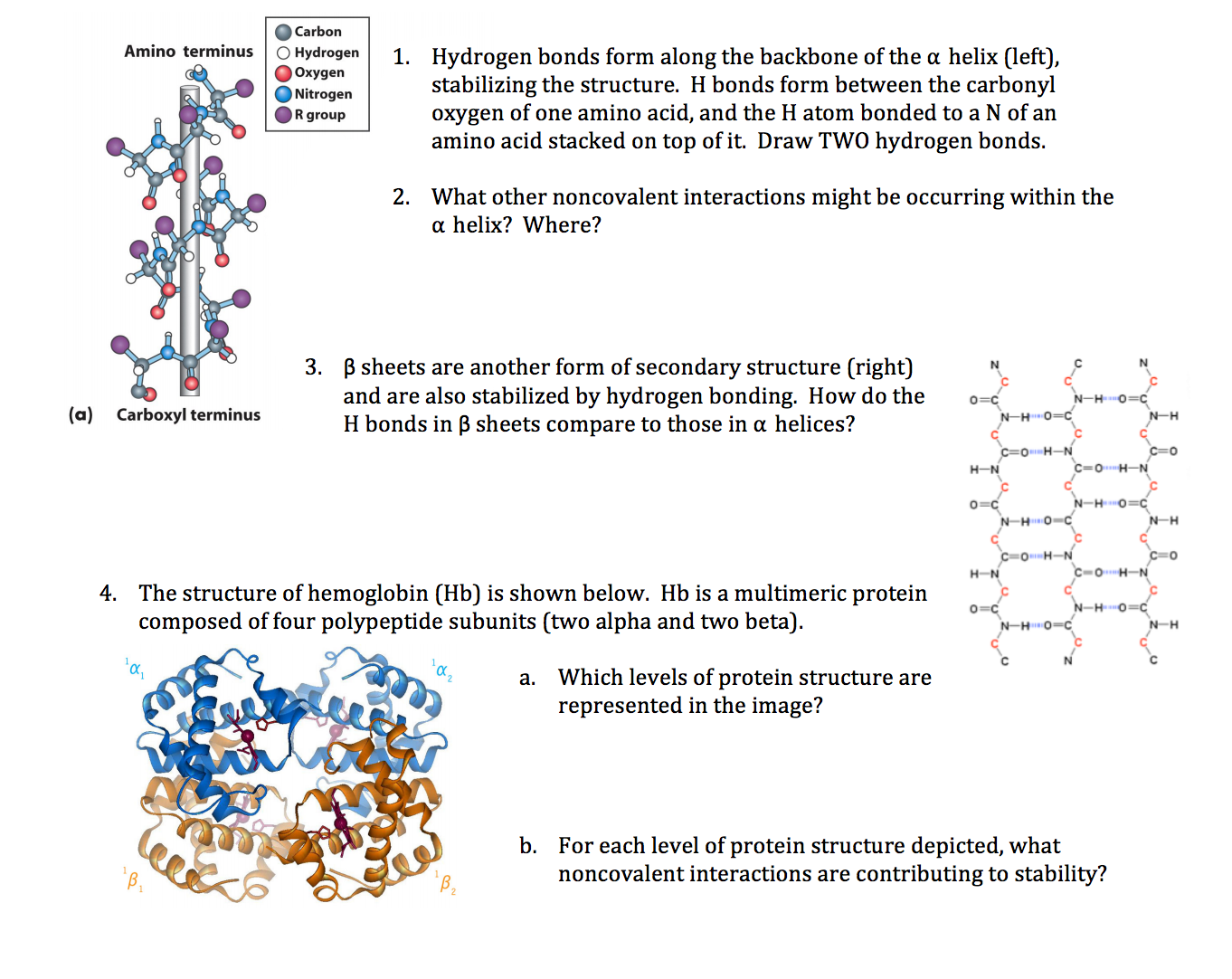
Think of an α-helix as more than just a spiral—it’s a tightly wound spring built by evolution for stability and function. Each turn contains about 3.6 amino acids, with hydrogen bonds forming between the backbone carbonyl oxygen of residue i and the amide hydrogen of residue i+4. This bonding pattern isn’t random; it’s a smart spatial solution that locks the backbone in place without wasting space. For more on the chemical interactions that keep these structures stable, see understanding the role of hydrogen bonds in stabilizing α-helices.
I remember my first time using PyMOL, zooming in on short helical segments that just looked like loops on the surface. The “aha” moment came when I specifically searched for those hydrogen bonds between residues four positions apart—suddenly, invisible threads lit up like neon signs showing me where the helix really was. For a complete overview of α-helix characteristics, see this complete overview of α-helix.
Why does this matter? Because these bonds create a rod-shaped cylinder about 5 Å wide, with each amino acid stepping roughly 1.5 Å along the axis. This compact architecture makes α-helices perfect for spanning membranes or serving as rigid scaffolds inside proteins—nature’s way of protecting polar backbone atoms from water without bulky side groups getting in the way. This structural role is especially important in membrane proteins; you can learn more about the functional importance of α-helices in membrane proteins.
One more thing: your brain loves patterns tied to function. Seeing α-helices as optimized springs—not just spirals—helps lock in understanding way better than memorizing numbers alone.
β-Sheets: Flat Beams Holding Things Together
Now flip that image: if α-helices are springs, β-sheets are flat architectural beams made from strands lined up side-by-side. Unlike helices, where hydrogen bonds happen within one chain (between residues close in sequence), β-sheet hydrogen bonds connect different strands—sometimes far apart in sequence but close in 3D space.
These strands can run parallel or antiparallel, which subtly changes bond angles and stability. The carbonyl oxygen on one strand reaches out across to bond with an amide hydrogen on another strand—a longer-range connection compared to helices.
A great example is lysozyme (129 amino acids): its β-sheets form stable “floorboards” holding catalytic sites steady, while surrounding α-helices act like walls or pillars adding rigidity and fine-tuning enzymatic activity.
Here’s a twist many beginners miss: α-helices rely on local, repetitive bonding inside one chain; β-sheets build bigger surfaces through cooperative bonding between chains. This difference is key to how proteins achieve complex 3D shapes.
Turns and Loops: Flexible Hinges with Purpose
Turns don’t follow neat repetitive patterns like helices or sheets—they’re short (usually 3-4 residues) flexible connectors that reverse chain direction sharply. Although less uniform, turns still use some hydrogen bonds for local stability.
Early on, I underestimated turns because they look messy—often lumped together as “random coils” in diagrams. But turns often carry critical biological roles: binding sites or catalytic loops often live here because flexibility allows dynamic interactions impossible for rigid helices or sheets.
For instance, I once studied a GPCR protein where a tiny loop between two transmembrane helices acted like a switch flipping during ligand binding—turns aren’t sloppy filler; they’re dynamic players essential for function.
Hydrogen Bonds Are Your Map — Learn How to Read Them
Programs like DSSP or STRIDE automatically assign secondary structures based on hydrogen bond geometry—typically around 2.8 Å distances for α-helices. But software can mislead if you don’t understand what those numbers mean physically: they reflect how tightly the backbone satisfies itself while balancing flexibility.
I mixed up 3_10 helices (i → i+3 bonds) with standard α-helices early on because both look spiral-ish. Only by focusing on bond distances and pitch (the rise per turn) did I reliably tell them apart—and believe me, this took several attempts!
A quick tip: our eyes want to categorize shapes visually first—that works sometimes but can fool you here. Instead, start by checking atomic-level bonding rules before trusting ribbon cartoons alone.
Why Do These Differences Even Matter?
You might be wondering why all this detail is worth it beyond exam questions? Because these structural differences shape how proteins behave:
- α-Helices shield polar backbone groups inside their cylinder—ideal for crossing oily membranes.
- β-Sheets create large surfaces that stabilize protein cores or sometimes cause nasty aggregations (think amyloid plaques).
- Turns provide flexibility needed for folding speed and transient interactions vital for signaling or catalysis.
Picture mistaking transmembrane α-helices in GPCRs for β-sheets—that would totally mess up models predicting how drugs access binding pockets! That kind of mistake is costly in drug discovery.
Common Pitfalls I Fell Into—and How You Can Dodge Them
My first big mistake was thinking all helices were alike—ignoring variants like π-helices (i → i+5 bonds) or 3_10 helices (i → i+3) which differ subtly but importantly in length per turn and stability.
What helped me?
- Zoom into atomic-level hydrogen bond networks using high-res crystal structures.
- Don’t rely only on ribbon views—they’re simplifications.
- Cross-check computational assignments with energy minimizations when possible.
- Practice spotting differences by comparing simple well-studied proteins like myoglobin or lysozyme using PyMOL alongside DSSP annotations.
Also watch out for loosely defined turns vs random coils especially when working with low-resolution data like circular dichroism spectra—here your biochemical intuition beats automated guesses every time.
A Simple Path Forward
If you’re starting out:
- Pick a small protein from the PDB (myoglobin is classic).
- Load it into PyMOL or Chimera.
- Focus specifically on identifying hydrogen bonds between backbone atoms.
- Look for i → i+4 bonds (α-helices) versus inter-strand bonds (β-sheets).
- Compare what you see with DSSP output.
- Repeat with different proteins until your eye catches these patterns naturally—not just shapes but bonding choreography underneath.
It takes weeks of practice—not months—to rewire your intuition this way, but trust me: once it clicks, recognizing secondary structures feels effortless rather than mysterious.
Wrapping Up
Distinguishing α-helices from other secondary structures isn’t just academic—it’s foundational to understanding proteins at every level—from enzymes speeding reactions to channels letting ions pass membranes to drugs targeting disease-causing proteins.
By making hydrogen bond patterns your compass—and appreciating their clever design—you’ll go from confused newbie to confident explorer faster than you expect. And that shift? That’s where true expertise begins.
So next time you face a jumble of ribbons and loops on your screen, ask yourself: What story do these invisible threads tell? Once you see the story clearly, everything else falls neatly into place.
Feel free to reach out if you want help walking through any specific examples or tools—I’ve been there! It’s tricky stuff at first, but worth every bit of head-scratching frustration along the way.